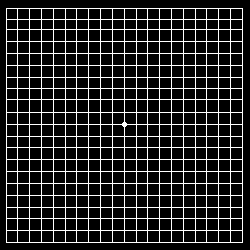
Normal Amsler grid


Download Amsler Grid (PDF File)
The Amsler grid, used since 1945, is a grid of horizontal and vertical lines used to monitor a person's central visual field. The grid was developed by Marc Amsler, a Swiss ophthalmologist. It is a diagnostic tool that aids in the detection of visual disturbances caused by changes in the retina, particularly the macula, such as age-related macular degeneration.
In the test, the person looks with each eye separately at the small dot in the center of the grid. Patients with macular disease may see wavy lines or some lines may be missing.
Amsler grids are often used to test one's vision at home. Patients with macular degeneration are often instructed to monitor their vision daily with an Amsler grid. This is because if their condition begins to worsen, treatment should be started immeadiately. Treatments for macular degeneration, such as Lucentis or Avastin, are more effective if they are started soon after disease progression occurs.
The original Amsler grid contained white lines on a black background, but today Amsler grids often have black lines on a white background. It is unclear whether one style is superior to the other.
Wear the eyeglasses that you normally wear for reading.
Position the chart 14 inches away from your face.
Cover one eye at a time with your hand.
Stare at the dot in the center. Do not let your eye drift from the center dot.
Contact your eye doctor immediately if:
Progress in the medical and surgical management of choroidal neovascularization (CNV) has lead to renewed interest in the field of macular function self-assessment. Earlier intervention, when CNV lesion size is smaller and visual acuity is better, improves the patient's visual outcome following any of the currently employed treatments for neovascular age-related macular degeneration (AMD). Since first introduced around 1950, the Amsler grid has been the primary method of patient self-assessment of the central visual field. However, the sensitivity of the Amsler grid in detecting vision defects has been documented to be approximately 50%, suggesting that approximately half of the scotomas that may be present are not detected. This had led to a search for more sensitive means for patients to evaluate their central vision for changes associated with choroidal neovascularization. Recently, a number of promising new technologies have emerged that may be employed by patients to monitor their macular function.
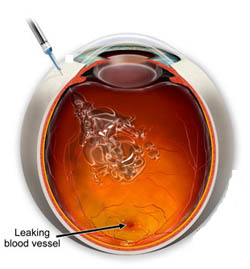
Antiangiogenic therapy has revolutionized the treatment of CNV. Today, many patients undergoing treatment for neovascular AMD will experience improvement in their vision. Ranibizumab (Lucentis) is one of the most widely used antiangiogenic drugs for the treatment of AMD. It is an antibody fragment that inhibits the activity of vascular endothelial growth factor (VEGF). When injected intravitreally, ranibizumab is associated with arrested growth of and leakage from CNV. In the MARINA study, a randomized double-masked study of the effectiveness of ranibizumab in AMD, both mean visual acuity and mean visual function scores showed improvement at 12 and 24 months. Similar improvements have been reported from the VISION study for pegaptanib (Macugen), another FDA-approved anti-VEGF medication. A recent systematic review of the literature found no statistically significant difference in the effectiveness of the two medications in halting the progression of neovascular AMD. A third VEGF inhibitor, bevacizumab (Avastin), is the most widely used drug employed in the treatment of neovascular AMD. It is not FDA-approved for the tretment of AMD, but is used "off label." The CATT study was a randomized controlled trial that compared the effectivness of Lucentis and Avastin. The study found no significant difference in the effectiveness of the two drugs. Aflibercept (Eylea) is the newest anti-VEGF drug that is used in the treatment of AMD. It has been shown to be as effective as the other agents, but has a longer duration of action so it does not need to be injected into the eye as frequently.
Earlier intervention, when neovascular lesion size is smaller and visual acuity is better, will generally improve the final visual outcome of therapy. A recent subgroup analysis of 24-month data from the MARINA study found that the two important predictors of final visual acuity were initial visual acuity and CNV lesion size. Similar analysis of the VISION data found earlier detection and treatment improved the visual outcome of Macugen-treated patients. Treatment of neovascular AMD patients earlier in the course of their disease with antiangiogenic agents results in greater vision recovery and, ultimately, less permanent vision loss. The earlier in the disease process that a patient presents for care, the greater the odds of avoiding severe vision loss.
The availability of highly effective therapies that can not only stabilize neovascular AMD, but can improve vision, has made it more important than ever to have patients with treatable lesions present for care as early in the disease process as possible, when vision is least affected. It has long been recognized that patient self-assessment of their vision on a daily basis offers the best hope of detecting disease progression in a timely manner. Yet, many patients with documented disease progression fail to detect changes while self monitoring their vision with the Amsler grid. This has lead to a call for making the development of a cost-effective self assessment tool for patients with macular degeneration a research priority.
There appears to be two primary reasons why home monitoring protocols utilizing the Amsler grid fails to detect disease progression: noncompliance and the subjective nature of the test. Noncompliance is frequently encountered with any health care regimen that patients are instructed to follow for prolonged periods of time, although the clinician can take steps that may maximize patient adherence and persistence. Objective tests of macular integrity, such as optical coherence tomography, require little or no patient input and could, at least in theory, be devised in such a way to screen for the presence of CNV. However, objective tests generally entail expensive instrumentation, making such tests impractical for patients to use at home. Subjective tests, such as the Amsler gird, are limited by the user's ability to understand the test, interpret their findings, and reliably report the results. The Amsler grid is an especially challenging test for patients because it requires the patient to delineate, on the chart, the perception of their vision defects, while fixating elsewhere. When compared to a gold standard of microperimetry performed with the scanning laser ophthalmoscope, the Amsler grid fails to detect approximately half of the scotomas that are present. Yet, because the Amsler grid test is inexpensive, easy to understand, and rapid to perform it is the most commonly employed method of macular function self-assessment.
A patient's subjective appreciation of their vision defects are influenced by a number of factors, including eccentric fixation, crowding effects, and the perceptual completion phenomena. Perceptual completion is a psychophysical phenomena wherein scotomas are filled-in with visual information from the rest of the visual field, thereby inhibiting subjective awareness of the vision defect. Because of these influences, only relatively large defects become subjectively apparent to the patient observing the Amsler grid. Technologies that are able to overcome or by-pass these influences, and thereby enhance subjective appreciation of vision defects, could prove valuable as a self-monitoring tool for patients with macular degeneration.
A number of alternatives have been developed in an effort to either improve or replace the Amsler gird. The Amsler grid has been modified in a variety of ways to improve its sensitivity, including red-on-black grids, low contrast grids, and computerized grids. Alternatives to the grid test include preferential hyperacuity perimetry, macular mapping test, and noise-field campimetry.